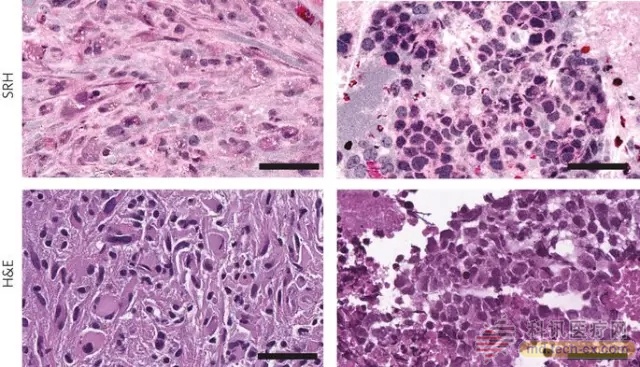
Release date: 2017-02-14 Recently, Nature Biomedical Engineering published a special issue of "Machine Learning", which introduced us to the wide application of machine learning in the medical field. We also launched the "Machine Learning" topic today to organize this information for readers. Now, let's take a look at the application of stimulated Raman scattering (Stimulated Raman Scattering, SRS) in brain tumor diagnosis and classification in areas such as artificial intelligence combined by it. The diagnosis and treatment of brain tumors depends on a number of factors, including clinical findings, neuroimaging studies depicting tumor location, and precise neuropathological diagnosis. The neuropathological academic diagnosis of tumor samples is critical for how neurosurgeons can adopt the best possible treatment strategy. Sample preparation of tissue sections involved in traditional methods of histopathological diagnosis generally follows the established fixation and staining protocols. This method has been practiced for decades and has yielded good results in accurately identifying tumor tissue. However, this approach has limitations: for example, the size of intraoperative tumor samples, whether using needle biopsy or core sampling, especially when considering the phenotypic heterogeneity of brain tumors, diagnostic capabilities may be affected . Furthermore, given the challenging and rarely observed histopathological features of rare brain tumors, they are likely to be misclassified. Finally, the processing of tumor samples may also introduce artifacts at the technical level. Reducing these sources of error is critical to maximizing accurate tumor diagnosis and can theoretically be achieved by applying intraoperative diagnostic tools for fresh specimens, resulting in reliable readout data and less processing time . ▲ clinically used SRS microscope (Source: "Nature Biomedical Engineering" ) In this regard, stimulated Raman scattering (SRS) has become a technique for accurately distinguishing different tissue characteristics based on the spectral properties of nucleotides, proteins, and lipids. SRS can be used to achieve rapid diagnosis of human skin cancer based on DNA properties and produce images comparable to traditional hematoxylin and eosin (H&E) formulations. In addition, the nature of SRS images, such as protein/lipid ratio, axon density, and cell number, can be used to identify invasive tumor cells in brain tissue with an accuracy of greater than 99%. Now, through the use of machine learning, Dr. Daniel Orringer and colleagues report in the journal Nature Biomedical Engineering the first histological image using the SRS microscope for rapid diagnosis and subclassification of brain tumors. This method is called stimulated Raman histology (SRH) and uses a specially designed SRS microscope in the operating room. Typically, two Raman shifts (~2845 cm-1 and ~2930 cm-1) are the basis for SRS imaging. The 2845 cm-1 shifted image highlights the lipids in the tissue sections and subtracts the above fraction from the 2930 cm-1 shift to show the structure of the protein and nucleotides. These features are sufficient to provide a clear image of tumor infiltration and tumor boundaries by assigning the image to two different colors. Dr. Orringer and colleagues used the opposite pseudo-coloring to produce H&E-like images, which is more familiar to pathologists. ▲ Images of stimulated Raman histology (SRH) and conventional hematoxylin and eosin (H&E) histology in patients with brain tissue specimens (Source: Nature) Dr. Orringer and his collaborators used SRH to scan samples from 101 neurosurgical patients, paying particular attention to the unique cellular and morphological properties of normal and tumor tissues. The reference tissue that examined the histological features of normal brain parenchyma was taken from the anterior temporal lobe after temporal lobe resection. SRH shows many unique features, including axons, lipofuscin particles and macrophage infiltration, which can sometimes affect diagnosis. For gliomas, SRH shows significant differences between low-grade and high-grade tumors, particularly in terms of cell size, nucleus shape, microvascular proliferation, and perinuclear halo. SRH can also be used to measure non-glial-derived tissues such as meningiomas and lymphomas. It is well known that gliomas are histopathologically heterogeneous such that a single small biopsy for gliomas may not be sufficient to provide a clear picture for diagnosis. In a low-grade glioma sample, Dr. Orringer and colleagues demonstrated the histological pattern of diffuse cells with high cytoplasmic density, but also showed the domain of hypercells and mitotically active cells showing anaplastic properties. Due to the rapid turnaround time of SRH, this approach allows neuropathologists to investigate different parts of the tumor in order to obtain a more comprehensive view of different cell types and cell behavior. Given that traditional methods of histopathological diagnosis involve H&E staining of frozen tissue sections, Dr. Orringer and co-authors must verify that the quality of the images produced by SRH is comparable to previous methods, providing neuropathologists with similar, rather than enhanced, information. The authors performed a quantitative analysis of the diagnosis performed by three neuropathologists who examined SRH data. Interestingly, neuropathologists can achieve 98% accuracy in distinguishing between diseased and non-lesional tissues, as well as nearly 100% accuracy of gels from non-glial tumors. In addition, when predicting the final diagnosis, there is near-perfect consistency between the observed traditional H&E diagnostic method and SRH, with an accuracy of 92% . With ease of use and fast processing times as short as 2.5 minutes, SRH is promising as a diagnostic tool for clinical implementation. The advantages of real-time feedback for fresh tissue are made, making SRH a valuable asset for neurosurgeons during surgery and can help determine the optimal treatment options for patients after resection. Conventional Raman spectroscopy using intraoperative Raman probes allows real-time measurement of the sample by contact with tissue prior to surgical resection, as well as spectral readings that can be correlated with cancerous or normal tissue. In the future, combining SRS and probe-based systems can provide diagnostic information and influence surgical decisions. Although the utility and safety of SRS and SRH needs to be validated by clinical trials, it is clear that Raman spectroscopy has shown great potential for improving diagnostic and therapeutic efficacy in neurosurgery . Reference materials: [1] Diagnostic imaging: Intraoperative virtual histology [2] Rapid intraoperative histology of unprocessed surgical specimens via fibre-laser-based stimulated Raman scattering microscopy Source: WuXi PharmaTech Zhoushan City Shuangying Aquatic Products Co., Ltd.  , https://www.shuangying-aquatic.com