Release date: 2017-01-05 Currently, the level of CA-125 protein in the blood is mainly used to measure the therapeutic response in women after receiving highly serous ovarian cancer (HGSOC) chemotherapy. However, after one or two cycles of chemotherapy, the CA-125 level did not change quickly enough to guide further treatment. In this new study, the researchers measured ctDNA levels that carried the gene TP53 mutation: the result was 99% of HGSOC patients. A total of 318 blood samples from 40 HGSOC patients were analyzed before, during, and after standard care treatment. CT images of patient tumors were also collected, including other data related to cancer progression. Source: Medical Valley Unwrinkled Smooth Film Dressing Frame,I.V. Transparent Dressing,Record Label Transparent Film Dressing,Breathable Waterproof Transparent Dressing Zhende Medical Co.,Ltd , https://www.zhendemedicals.com
Recently, a retrospective study of blood samples from cancer patients was published in the journal PLOS Medicine. The data show that circulating tumor DNA (ctDNA) levels detected in blood tests are associated with the size of ovarian cancer and can predict patient treatment. The degree of response or the corresponding time of disease progression. The researchers who revealed this important discovery were Dr. Nitzan Rosenfeld and Dr. James Brenton from Cancer Research UK in Cambridge, England, and their colleagues. 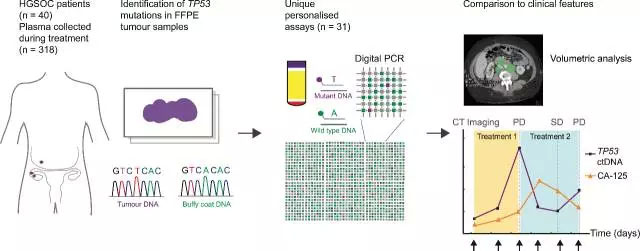
Circulating tumor DNA analysis process
The primary outcome of the study was that the level of TP53 mutation (TP53MAF) in ctDNA was correlated with tumor volume measured by CT scan (Pearson r = 0.59, p < 0.001); pre-treatment tp53maf levels were also associated with progression in each patient. Time related. The change in the level of ca-125 takes 84 days to reflect the completeness after chemotherapy, whereas the change in tp53maf requires only a median of 37 days to reflect this goal. In the treatment of recurrent patients, when the reduction in tp53maf exceeds 60%, it is associated with a longer progression time, while a reduction of less than 60% is associated with adverse effects of chemotherapy, and the progression time is less than 6 months. It appears that the study is limited by the small sample size, the heterogeneity of the treatment regimen, and the retrospective nature of the experimental design. In the long run, scientists need to conduct more prospective trials involving more subjects and uniform treatment options to further confirm the results. 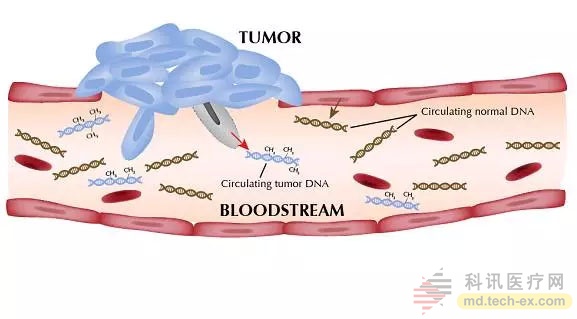
Circulating tumor DNA in the blood
The researchers believe that because the DNA in plasma is generally easy to measure, and the low cost and high speed of ctDNA testing, these findings will have strong clinical potential. Obtaining very early response information will help patients and physicians to consider alternative treatment options and is highly useful in clinical trials linking biomarkers to targeted therapies.