Professor Shi Chunmeng of the Third Military Medical University of Chongqing published a document entitled "Identification of a fluorescent small-molecule enhancer for therapeutic autophagy in colorectal cancer by targeting mitochondrial protein translocase TIM44" on November 14, 2016. The article, for the first time identified a new near-infrared (NIR) fluorophore autophagy enhancer can be used as a potential colorectal cancer targeted drug, let's take a look. Sleep improvement/Anti-anxiety We're well-known as one of the leading Health care supplement--Sleep improvement/Anti-anxiety manufacturers and suppliers for our quality products and good service. Please feel free to buy or wholesale bulk 5-htp Griffonia Seed Powder, L-Theanine powder, Valeriana Officinalis Acid Root Extract Powder, Apigenin and nicotinamide mononucleotide 99% nmn powder for sale at the best price from our factory. Anti-Anxiety Plant Extract,Sleep Improvement Powder,Sleep Improvement Extract,Sleep Improvement Xi'an Natural Field Bio-Technique Co., Ltd. , https://www.natural-field.com
Â
OBJECTIVE: Since the regulation of autophagy is beneficial for cancer treatment, it is important to identify novel autophagy enhancers. However, current autophagy-inducing anticancer agents exert side effects due to their non-specific biodistribution in off-target tissues. This study aimed to develop a multifunctional reagent to integrate cancer targeting, imaging and treatment, and to investigate its mechanism.
Â
Design: Synthesis, screening, and identification of a series of near-infrared (NIR) fluorophores targeting mitochondria that enhance autophagy activity and test optical and biological effects in vitro and in vivo. The underlying mechanisms of research using inhibitors, small interfering RNA (siRNA), RNA sequencing, mass spectrometry and human samples.
Â
RESULTS: The investigators screened and identified a novel NIR autophagy enhancer, IR-58, which exhibited significant tumor-selective killing.
Â
1. IR-58 synthesis pathway 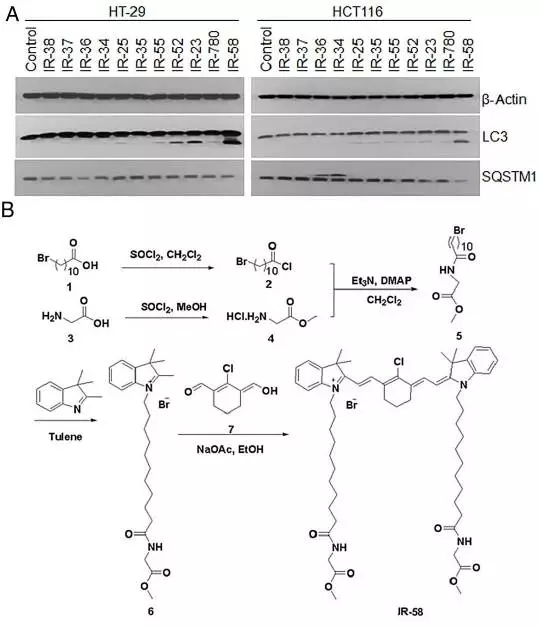
Figure 1 IR-58 is capable of inducing autophagy in CRC cells (Fig. 1A). The chemical synthesis method of IR-58 is shown in Figure 1B. After the synthesis of compounds 2 and 4, a key intermediate (compound 5, yield 60%) was synthesized by amidation of compounds 2 and 4 under the catalysis of Et 3 N. The chemical structure of Compound 5 was measured using 1H-NMR (1H-NMR). Subsequently, compound 6 in the form of a quaternary ammonium salt is prepared by N-alkylation of 2,3,3-trimethyl-3H oxime and compound 5 in 1,2-dichlorobenzene. Finally, compound 6 was reacted with compound 7 to synthesize IR-58 (1.63 g, yield 30%, green viscous solid) by condensation using sodium acetate as a catalyst.
Â
2. In vivo function study of IR-58 cells and their animals
The investigators studied the potential of IR-58 over tumor enrichment in HT-29 and HCT116 CRC xenografts. Human HT-29 and HCT116 CRC xenografts were implanted subcutaneously in nude mice, and were imaged after a single intravenous injection of IR-58. 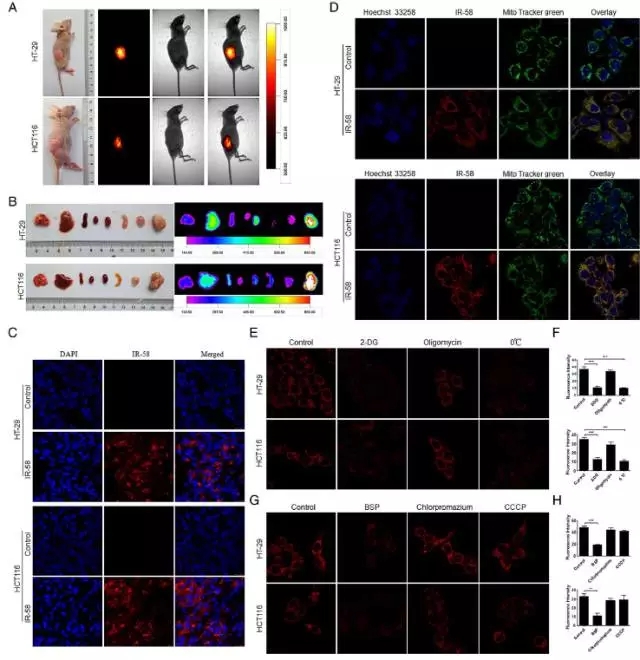
Figure 2 A, B shows that the transplanted tumor showed strong fluorescence after injection of IR-58. It was confirmed by histopathological analysis that IR-58 iodide was enriched in tumor cell solute. As determined by subcellular localization experiments, IR-58 was only accumulated in the mitochondria of tumor cells (Panel D). Since mitochondria are the energy center of cells and IR-58 is located in mitochondria, the researchers speculate that IR-58 uptake is energy-dependent and confirmed. (Figures E, F). Pretreatment of HT-29 and HCT116 cells with 2-deoxy-d-glucose (2-DG; glycolytic inhibitor) and oligomycin (oxidative phosphorylation inhibitor) revealed that 2-DG significantly inhibited IR-58 Uptake indicated that the uptake of IR-58 was glycolytically dependent, whereas oligomycin did not. (Figures E, F). When HT-29 and HCT116 cells were pretreated with sulfosyl bromide (BSP, a competitive inhibitor of OATP transporter), the fluorescence value was significantly lower than that of untreated cells, indicating that IR-58 uptake was through organic anions. Transporter polypeptide (OATP) mediated (Figure G, H).
Â
3. Research on the mechanism of action of IR-58
Â
IR-58 is preferentially enriched in mitochondria of colorectal cancer (CRC) cells and xenografts, a glycolytic-dependent and organic anion transporter polypeptide-dependent process. IR-58 kills tumor cells and induces apoptosis by inducing excessive autophagy, which is mediated through the reactive oxygen species (ROS)-Akt-mammalian rapamycin target protein (mTOR) pathway. RNA sequencing, mass spectrometry and siRNA interference studies indicate that inhibition of the mitochondrial inner membrane 44 (TIM44)-superoxide dismutase 2 (SOD2) pathway transposase results in IR-58-induced excessive ROS, autophagy and apoptosis. TIM44 expression was positively correlated with CRC development and poor patient prognosis. 
Figure 3 HT-29, HCT116, A549 cells were treated with 10 μM IR-58 for 24 hours and a blank control was set up, followed by RNA sequencing and mass spectrometry to increase mRNA expression of overlapping genes. 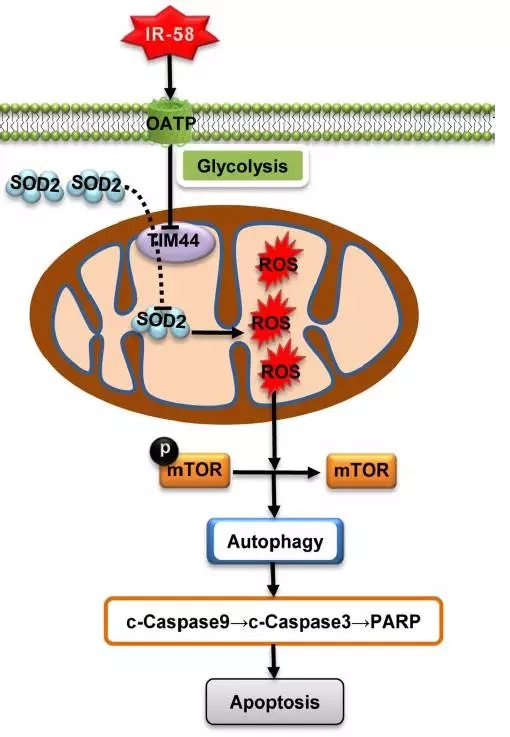
Figure 4 shows the proposed model of IR-58 autophagy induction mechanism. IR-58 is preferentially enriched in mitochondria of tumor cells in an organic anion transport polypeptide (OATP)-dependent and glycolytic-dependent manner. IR-58 significantly inhibits the expression of 44 (TIM44) translocator in mitochondrial membrane, resulting in a decrease in the uptake of superoxide dismutase 2 (SOD2) into mitochondria, resulting in excessive production of reactive oxygen species (ROS) and mammalian rapamycin. Dephosphorylation of the target protein (mTOR) triggers autophagy. Subsequently, endogenous apoptosis is triggered. PARP, poly ADP-ribose polymerase.
Conclusion: A novel NIR small molecule autophagy enhancer, IR-58, has been developed with mitochondrial targeted imaging and therapeutic capabilities for CRC therapy. Furthermore, for the first time, TIM44 was identified as a potential oncogene that plays an important role in autophagy via the TIM44-SOD2-ROS-mTOR pathway.
Â
New findings: IR-58 was identified as a novel tumor autophagy-inducing small molecule dye that preferentially enriches mitochondria in tumor cells and specific target tumor sites in vivo.
Â
How does it affect clinical practice for the foreseeable future?
IR-58 may represent a novel autophagy-induced tumoricidal small molecule enhancer, and the TIM44-SOD2-ROS-mTOR pathway may be a potential target for clinical CRC therapy. This work may also provide a potential strategy to develop multifunctional small molecule reagents for imaging-guided CRC treatment, which can enhance the specificity of tumor targeting and reduce potential side effects in isolated target tissues, as well as enable real-time monitoring of tumors. Response to treatment.
Â
Original: Huang Y, Zhou J, Luo S, et al. Identification of a fluorescent small-molecule enhancer for therapeutic autophagy in colorectal cancer by targeting mitochondrial protein translocase TIM44[J]. Gut , 2016: gutjnl-2016-311909.
Â
The RNA Sequencing used in this study was provided by Ruibo Bio, and the animal experiments involved as well as siRNA Ruibo were provided.
Â
Related meeting recommendations
2017 4th non-coding RNA and epigenetic research experience exchange meeting
Meeting time: 2017.6.10 -6.11 Meeting place: Guangzhou Science City
Conference details: http://
Gut: IR-58 - Potential colorectal cancer targeted drug