Release date: 2017-06-12 In the in vitro diagnostic industry (IVD), immediate detection (POCT) is an important part of laboratory medicine. Because of its small size, convenient carrying, easy operation, and rapid clinical results, it has developed rapidly in the application of clinical departments. The average annual growth rate is as high as 30%. Â Â Â Â Â Â Source: Kexun Medical Network Tetanus Vaccine,Hepatitis B Vaccine For Adults,Tetanus Booster,Td Vaccine FOSHAN PHARMA CO., LTD. , https://www.forepharm.com
In the hospital, POCT is mainly used in clinical departments such as emergency department, ICU, respiratory department, cardiology department, and operating room. This technology complements traditional testing in terms of timeliness and flexibility, and also compensates for the lack of testing resources in primary hospitals.
Â
In the POCT immunoassay device, the instrument detection method is mostly based on the most basic double antibody sandwich method, and in order to realize such immunoassay, a marker, a carrier, a reaction control device, and the like are required.
Immunolabeling technology: the advantages and disadvantages of markers
The colloidal gold technology developed in the 1980s (for example, early pregnancy test strips) has been used since its simplicity, convenience and speed. However, it can only achieve a qualitative diagnosis and cannot meet the quantitative needs of doctors.
Subsequent substances with fluorescent materials, such as Rhodamine, which is the most widely used, and up-lighting materials, lanthanides, quantum dots, etc., which have been used in recent years, are also the most used marker categories for POCT manufacturers. However, the blood sample itself produces fluorescence, which is interfered by incident light scattering and background fluorescence, so the sensitivity and quantitative analysis capabilities of the method are limited by noise.
In order to improve the shortcomings of fluorescent materials and achieve strong anti-interference ability and accurate detection, "Nanomagnetic beads" has become a new marker choice for POCT manufacturers. Because the blood sample itself does not have a magnetic signal, the nano-magnetic beads have no problem of interference with fluorescent background noise, and also provide a new way for more sensitive detection.
According to the type of the marker, it can be divided into: Serial number Marker type representative 1 Colored microspheres Latex microspheres, colloidal gold, colloidal carbon, colloidal selenium 2 Fluorescent material Rhodamine, upturn luminescent materials, lanthanides, quantum dots 3 Magnetic microsphere Super-smooth nanobeads
Immunochromatography: replacement of vectors
In the use of the carrier for the immunoreaction, the most widely used is immunochromatography, and a nitrocellulose membrane (i.e., NC membrane) is used as a carrier. Due to the uneven and inconsistent structure of the NC film, the materials are thin and thick, and the degree of density is different, which makes the sample test results less consistent on different test cards. In addition, the principle of the chromatographic technique is the capillary action of the liquid. The velocity, sample size, reaction time, etc. of the sample to be tested are uncontrollable and will vary with the sample. Other reagents (fluorescent substances, secondary antibodies, etc.) involved in the test reaction also change. All of the above will result in inaccurate test results. In extreme cases, very viscous samples can't even flow, and the test fails completely. 
In order to improve the problem on the chromatographic membrane, a technique of replacing the NC membrane with a capillary has appeared, using a microfluidic channel as a carrier. For example, the Triage test card of Alere Corporation of the United States, its test time is not fixed, the main reason is the difference between the samples, the test results are affected by the following factors:
Although it is called "microfluidic" technology, it is actually an uncontrolled natural process. The flow rate of reagents is still affected by many factors, and the results are still uncontrollable. The card is stored refrigerated, and the test card needs to be taken out before rewarming before use, otherwise the temperature will affect the measurement results. 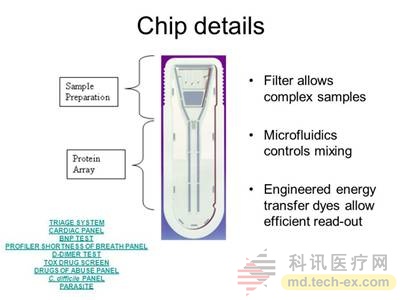
New technology update: m16 magnetic sensitivity immunoassay was born
In order to break through the shortcomings of the chromatographic principle, it is only possible to abandon the immunochromatography technology and find new methods and technologies.
As early as 1998, the US Naval Laboratory proposed the idea of ​​using GMR (giant magnetoresistance (GMR) effect and immunomagnetic labeling to realize GMR biosensor. They have demonstrated the feasibility of GMR sensors in the field of bioassay by measuring DNA , antigen-antibody, donor and receptor experiments, which has opened the curtain of magnetic-sensitive immunoassay technology.
Â
In addition to the US Naval Laboratory, NVE in the United States, Stanford University in the United States, University of Bielefeld in Germany, and University of Lisbon in Portugal also conduct research on the application of GMR biosensors for magnetic-sensitive immunoassays; the Chinese Academy of Sciences and Tsinghua University University, University of Electronic Science and Technology, etc., but these units have not made GMR technology product.
Magnetic sensitivity immunoassay combines biotechnology, semiconductor technology, magnetic thin film technology and weak signal detection technology. Through the detection of immunomagnetic markers, the composition of the test solution and the concentration of the components to be tested can be accurately determined.
M16 magnetic sensitivity immunoassay advantage
Shenzhen Libang's POCT product m16 magnetic-sensitive immunoassay analyzer (hereinafter referred to as m16) successfully commercialized magnetic-sensitive immunoassay technology. It uses the most popular biochip as the carrier, and creatively combines the GMR-based biochip with active intelligent microfluidics and nanobead marking technology. This is a major breakthrough in the history of POCT, effectively solving the problem of immune detection susceptible to interference.
     The use of microarray biochips enables m16 to be calibrated in real time, correcting differences between batches, changes in transport and storage conditions, and differences in test environments, resulting in significant improvements in sensitivity and precision. Its biggest advantage is the joint inspection of more than ten or even more than 30 projects. 
      The unique microfluidic technology is actively controlled by pumps and valves in the instrument. Different passive microfluids on the market can accurately control the temperature, flow rate, flow rate, flow direction and reaction time of various reagents involved in the reaction. The result is more precise. 
The nano magnetic bead particle technology as a marker can greatly improve the detection sensitivity and realize the detection of a single biomolecule in the m16 detection system. 
In summary, POCT has been in development for more than 20 years, and has experienced several major technological breakthroughs. The popularization of new technologies requires a long process. In the near future, a new generation of POCT technology-magnetic sensitivity immunoassay technology will shine.