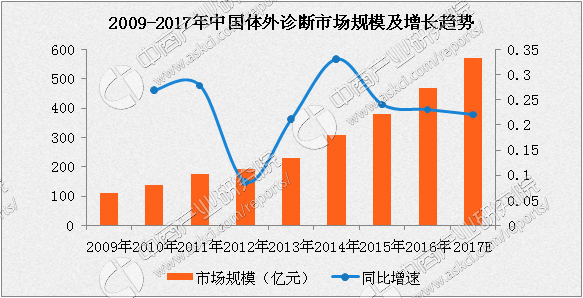
In vitro diagnosis, that is, IVD, refers to products and services that obtain clinical diagnostic information by detecting blood, body fluids, tissues and other samples outside the human body, and then determine the disease or body function. The in vitro diagnostic products are controlled by corresponding instruments. Reagents constitute a complete system, including diagnostic instruments, diagnostic reagents and related calibration quality control peripherals. About 80% of clinical diagnostic information comes from in vitro diagnosis, and in vitro diagnosis has become an indispensable medical tool for human disease prevention, diagnosis and treatment. In vitro diagnostic products do not directly affect the human body. According to the detection principle or detection method, they are mainly divided into biochemical diagnosis, immunodiagnosis, molecular diagnosis, microbial diagnosis, urine diagnosis, coagulation diagnosis, etc., among which clinical biochemistry, immunodiagnosis and molecular diagnosis are At present, the main technology of in vitro diagnosis in China. China's in vitro diagnostic industry market size and forecast The in vitro diagnostic industry started late in China, and domestic innovation technology and overall medical level are not as advanced as foreign countries. In 2009, the scale of China's in vitro diagnostic market is still relatively small, only 10.8 billion yuan. In recent years, with the gradual improvement and support of domestic in vitro diagnostic policies and the increasing demand in the medical market, the reputation of in vitro diagnostic technologies and products has made great progress. At present, according to the data of the China Pharmaceutical Industry Information Center, as of 2014, the in vitro diagnostic market reached 30.6 billion yuan, and in 2013 it was 23 billion yuan, a year-on-year growth rate of 33.04%. In addition, the China Business Research Institute "2016-2021 Global and China In Vitro Diagnostic Industry Market Research Report" predicts that the domestic in vitro diagnostic market scale will increase by 22%-25% in 2015-2017, and is expected to be domestic in 2017. The in vitro market size will reach 56.9 billion yuan, a year-on-year increase of 22%. Source: China Business Research Institute Pain Relief Patch(Pain Areas)
Pain Relief Patch
Pain Relief Patch(Pain Areas),Shouler Pain Relief Patch,Joints Pain Relief Patch, Muscle Pain Relief Patch Shandong XiJieYiTong International Trade Co.,Ltd. , https://www.xjplaster.com
[Name] Medical Cold Patch
[Package Dimension] 6cm×8cm 4pieces/box
The pain relief patch is composed of three layers, namely, backing lining, middle gel and protective film. It is free from pharmacological, immunological or metabolic ingredients.
[Scope of Application]
For cold physiotherapy, closed soft tissue only.
[Indications]
The patches give fast acting pain relief for strains, sprains, cramp, bruises, swollen areas or joint stiffness.
[How To Use a Patch]
Please follow the Schematic Diagram. One piece, one time.
The curing effect of each piece can last for 6-8 hours.
[Attention]
Do not apply the patch on the problematic skin, such as wounds, eczema, dermatitis,or in the eyes. People allergic to herbs and the pregnant are advised not to use the medication. If swelling or irritation occurs, please stop using and if any of these effects persist or worsen.notify your doctor or pharmacist promptly. Children using the patch must be supervised by adults.
[Storage Conditions]
Store below 30c in a dry place away from heat and direct sunlight.